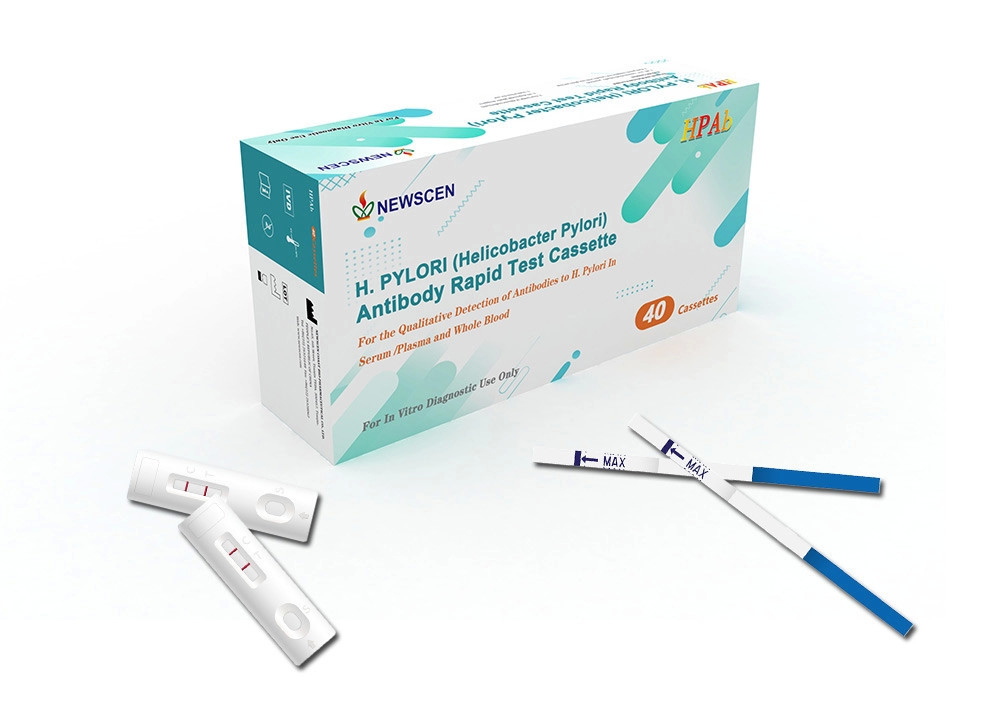
W5 Minutes Home 100ul Plasma H Pylori Test Kit FDA CE Approved
Features
High Sensitivity
High Specificity
Reliable: high accurate, early detection of the presence of H.Pylori
Simple: No Instrument Required
Convenient: Room Temperature Storage, Built-In Control line
Fast: Results in 5-30 minutes, strong positive results may be observed promptly within 3 minutes
Certified by Authoritative Certification System and Standards
Winner of "the National Rapid Diagnostic Kit for Clinical Performance Assessment"
Principle
The H. pylori Rapid Test is a lateral flow, immunochromatographic-screening test. H. pylori antigens are precoated onto membrane as a capture reagent on the test band region. During the assay the specimen serum is allowed to react with H. pylori antigens-gold conjugate.
The mixture then moves laterally on the membrane chromatographically to the test region with immobilized antigens of H. pylori. If H. pylori antibodies are present in the specimen, two colored bands are formed on the test region. One colored band in the test region indicates a negative result.

Reagents and Materials Provided
1. One pouched cassette with desiccant.
2. One bottle of diluent buffer.
3. Instruction for use.
Materials Required But Not Provided
1. Clock or timer
2. Specimen collection tube
Warning and Precautions
1. For in vitro diagnostic use only.
2. Do not interchange reagents from different lots or use test kit beyond expiration date.
3. All patient samples should be treated as if capable of transmitting diseases.
4. Icteric, lipemic, hemolysis, heat treated and contaminated sera may cause erroneous results.

Assay Procedure
Serum/Plasma
Add 70-100 ul or 2-3 drops serum or plasma into sample well. Observe the result in 10-15 minutes.
Whole blood
Add 1 drop of whole blood into sample well, after all blood completely absorbed, add 1 drop of whole blood diluent. Observe the result in 10-15 minutes. add 1 drop of diluent. Observe the result in 5-15 minutes.


Negative: Only one pink band appears. This indicates that no H. pylori antibodies have been detected.
Positive: Two pink bands appear. This indicates that the specimen contains H. pylori antibodies.
Invalid: If no band appears, this indicates a possible error in performing the test. The test should be repeated using a new device.
Storage
Store the test kits at temperature 4-30°C,in the sealed pouch for the duration of the shelf life (24 months).
Limitation of The Procedure
1. The test is to be used for the qualitative detection of antibodies to H. pylori.
2. A negative result does not rule out infection by H. pylori because the antibodies to H. pylori may be absent or may not be present in sufficient quality to be detected at early stage of infection.


 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!